Do terms like lye concentration, water discounting and water/lye ratio make your head spin? Hopefully not anymore after reading this post.
Getting a good grasp on how much water to use in a cold process soap recipe will take your soap making to the next level. It will ultimately enable you to come up with your own soap recipes and tailor them to exactly the soap project you have in mind.

Jump to:
When I first started soap making I strictly adhered to soap recipes I found on reputable sites. But after some time and after taking a soap formulating workshop I started to pick apart every recipe I came across: Why this amount of water? Why this amount of lye? Why this profile of fatty acids? And so on.
I experimented with recipes a lot and particularly with lye concentrations. Playing around with numbers on a lye calculator and coming up with perfect soap recipes is great fun to me. And over the years I've gotten very familiar with it.
But in the beginning it felt quite daunting. So, if you're new to all of this, don't fret I will attempt to break it down for you.
Why is Water used in Soap Making?
In order to make soap oils/fats need to be mixed with lye (sodium hydroxide or caustic soda). One is acidic the other alkaline. When the two come together they start what's called the saponification process. Lye molecules will latch on to fat molecules and turn them into soap.
Lye has to be dissolved in water before we can use it to make soap. A lye solution is created by measuring out an amount of distilled water and stirring in an exact amount of lye until fully dissolved. This process has to happen under proper safety precautions. Sodium Hydroxide is a chemical substance that can cause skin burns and severe injury to eyes and should always be handled with care.
The water itself doesn't undergo any molecular changes in the soap making process. It enters as H2o it exits as H2o. It's simply a vehicle. Most of it will evaporate from the bar of soap during the drying period or "cure time".
Different ways to measure water content
When you open up Soapcalc the most widely used soap recipe calculator, you will find three options at the top to calculate the water content:
- Water as percentage of oil
- Lye concentration
- Water : Lye ratio
This leads to a lot of misunderstandings and confusion amongst soap makers. Today we're talking about lye concentration which is the most precise way to measure water content. If you want to dive deep into what each of these terms means and why it's best to use lye concentration as a measurement , read this article on water discount.
Lye Discount / Superfat
Different oils/fats need different amounts of lye to turn into soap. For example, in order to turn 1 ounce of coconut oil into soap 0.18 ounce of lye are needed, but for 1 ounce of olive oil only 0.14 ounce are needed. These different saponification values are what lye calculators use to calculate the right amount of lye needed in a soap recipe.
Now, most handmade soap recipes will use less lye than needed in order to have some unsaponified or non-altered oil molecules present. This method is called super fatting (or lye discount) and results in a gentle and nourishing bar of soap.
Too much lye will result in a lye heavy soap that isn't safe to use. Molecules of lye that had no fat molecule to latch onto will still be present in the soap causing skin irritations or burns. Too little lye will result in oil pooling on top of the soap or in a very soft bar that never hardens.
That's why it's important to get the lye amount right, to run every soap recipe through a soap calculator and to be precise when weighing.
Let's do an example together:
I want to turn 20 ounces of olive oil into soap.
At a Super Fat of 0% (meaning no extra oil will be left in the finished soap), the amount of lye to use would be 2.71 ounces (76g).
At a Super Fat of 8% (meaning 8% of the oils will stay untouched), the amount of would be 2.49 ounces of lye (70g). By decreasing the amount of lye (Lye Discount) the soap contains more free oil molecules (Super Fat).
Let's go with a Super Fat of 5% for this example, which would be 2.57 ounces (73g) of lye. From here on out the amount of lye will stay the same. We know that it will produce a bar of soap gentle and safe to use.
Different Lye Concentrations
Continuing with our example, lets look at different lye concentrations. Remember the amount of lye will not change. However, depending on how much water is added, the lye concentration will change. Typically in soap making, lye concentrations between 25% (full water) and 40% (high water discount) are most commonly used. Using the number from the example above, let's look at how the water amount changes at 33%, 37% and 40% lye concentration.
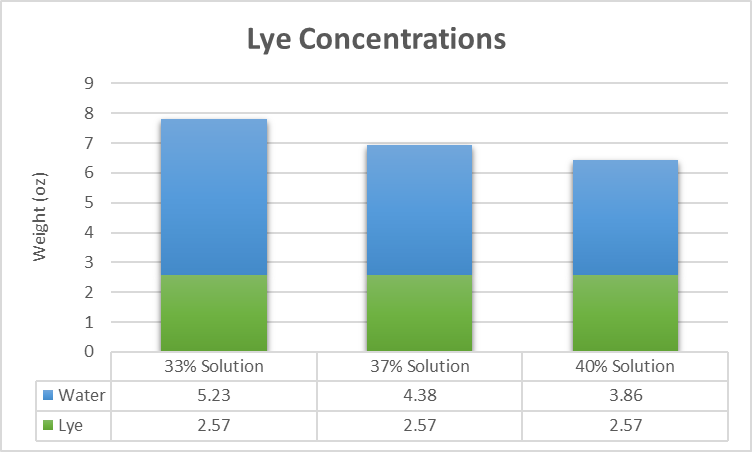
As you can see the amount of water needed for each lye-water solution varies quite a lot especially looking at the difference between 33% and 40% of about 1.3 ounces. Now, what difference does it make in the finished soap whether you use more or less water? Didn't I say earlier it's only a vehicle and will evaporate any way? It matters for different reasons.
Most beginner cold process recipes will use a lye concentration of 33%. There's one good reason why you should use this water content for your first batch of soap: it gives you more time to work.
The soap making process is a race against the clock. The minute lye molecules and fat molecules begin their dance your soap batter will begin to solidify. Sometimes faster, sometimes slower. Adding more water in the beginning will allow you to work slower - a big plus when learning a new skill!
Benefits of Using a Water Discount
After you have a couple of batches under your belt there isn't much benefit in my opinion to use such a high water content for soaping. As a matter of fact using a lye concentration between 37-40% or can have many advantages, as we will see here:
- helps reduces soda ash (caustic soda)
- reduces chance of glycerin rivers
- makes unmolding soap from silicone molds easier
- helps soap go into gel phase faster and at a higher temperature
Soaping with a 40% lye concentration or higher becomes necessary if you're planning to add fresh ingredients, like avocado puree or additional liquids, like heavy cream at trace. That way you don't end up with too much liquid in your finished bar.
Reasons to Use More Water
As I mentioned briefly before, one of the big advantages of using more water (a lye concentration of around 33%) is that your soap will trace slower. This will give you more time to work, which is not only great if you're a beginner, but also if your attempting to do an intricate soap design.
Additionally, if you're planning to use ingredients that will add additional sugar, like honey or additives that will speed up trace, like beeswax or certain fragrance oils, you're better off using a higher amount of water.
Does a High Lye Concentration cut down on Cure Time?
That's exactly what I wanted to find out when I started a little experiment. I made two small batches of soap, one was made with a lye concentration of 33% the other with a lye concentration of 38%. Everything else was exactly the same, the amounts of oils, lye and fragrance. Both soaps went into the freezer right after pouring to prevent gelling.
Using a precise jewelry scale, I weight each bar over a period of 18 days. My goal was to track how much water evaporates from each bar over time. In order to determine if one cured faster than the other. Remember, a bar of soap is completely cured if there's no more change in weight, meaning it's fully dried.
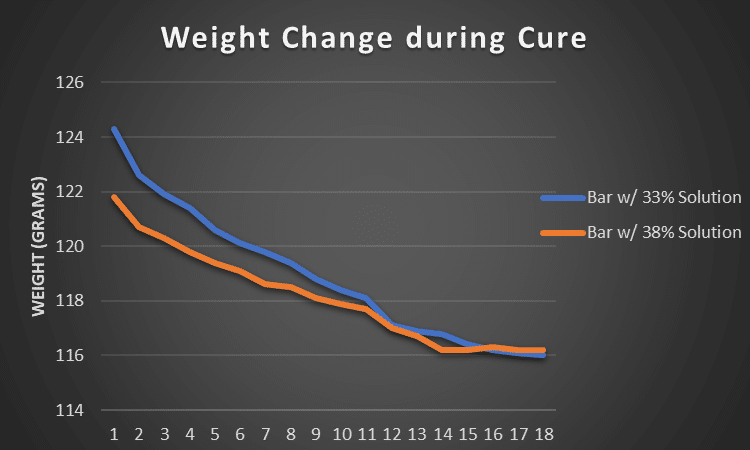
The 33% bar started out with more weight - no surprise here. But then over time it caught up with the 38% bar simply because the water loss was a lot steeper. Eventually after this initial period both bars moved along pretty evenly with small weight decreases.
This was just one little trial, but it's super interesting to see that first, the biggest decrease in weight happens in the first two weeks of curing. And secondly, a higher water discount might not automatically mean your soap will cure faster.
What's your experience with this? Let me know if you've made similar observations.
Until next time. Happy Tinkering!





cassie
thanks so much for this learned so much! so excited to start!